 While widespread knowledge of the health effects of the drug Accutane that is used to treat severe cases of acne didn’t occur until the late 1990s, the use of the remedy actually goes back to the 1930s.
While widespread knowledge of the health effects of the drug Accutane that is used to treat severe cases of acne didn’t occur until the late 1990s, the use of the remedy actually goes back to the 1930s.
Accutane is actually a form of Vitamin A that is designed to cause oily skin to dry and help skin renew itself more quickly. Accutane built upon knowledge pioneered decades ago that high dosages of Vitamin A were effective for skin conditions, given the vitamin’s fat soluble qualities. Fat soluble vitamins are stored by the body for much longer periods than water soluble vitamins, and this quality made high doses of Vitamin A an especially effective treatment for skin conditions.
Accutane was developed in 1982 by Hoffman-La Roche and its discovery led to several awards for Dr. Gary Peck, who received a U.S. patent for the drug and its treatment of acne. Almost from the start, the dosages needed for effective treatment were disputed. Although doctors commonly prescribed high dosages of the drug, technically known as isotretinoin, lower dosages were found to be just as effective for treatment of skin conditions.
Another key side effect of the drug was its impact on pregnant women. Despite widespread knowledge of Accutane’s ability to cause birth defects in up to 30 percent of patients compared to 3 to 5 percent for the population as a whole, in most cases doctors weren’t administering pregnancy tests to women before prescribing the drug.
Upon suspicion that the number of pregnant women taking the drug was being underreported, the U.S. Food & Drug Administration instituted a number of restrictions for prescribing the drug.
In 2002, Roche’s patent for isotretinoin expired, which led a number of competitors to develop generic versions. Roche stopped distributing and manufacturing the drug on June 29, 2009, although a unit of the company still manufactures and distributes it under the name Roaccutane in other parts of the world.
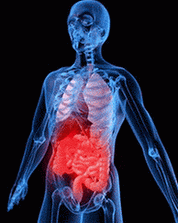 Accutane’s side effects can include ulcerative colitis
Accutane’s side effects can include ulcerative colitis/images/topic.png)


/images/contact-banner.jpg)